
The geometry of the CH4 molecule can then be determined using the Valence Shell Electron Pair Repulsion Theory (VSEPR Theory), which stipulates that molecules will adopt a CH4 geometrical formation that the electrons have from one another.įinally, you must calculate the C-H bond’s strength by adding their bond polarities (dipole moment properties of the CH4 molecule). The first stage is to sketch the CH4 molecule’s Lewis structure the second step is to determine the CH4 molecule’s geometry, and the final step is to determine the CH4 molecule’s bond polarities (dipole moment) and add them together.ĭrawing the CH4 Lewis structure is a method of representing a molecule using a diagram that shows the number of valence electrons and bond electron pairs in the CH4 molecule. 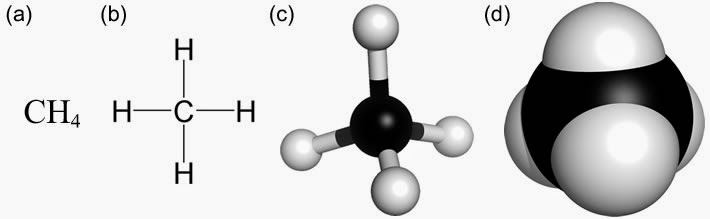
When attempting to determine the polarity of the CH4 molecule, a three-step procedure can be used. Key Points To Consider When Determining The Polarity of CH4 Molecule The chemical composition of the methane molecule is 4 hydrogen atoms and 1 carbon atom in the middle. Mol mass of CH4 = 1 * 12 (atomic mass of C) + 4 * 1(atomic mass of H) += 16.04 g The chemical equation of methane bio synthesis in the enzymatic reaction is shown below.ĬO2 + 4H2 -enzyme(methyl coenzyme M reductase)-> CH4 + 2H2O Preparation of methane(CH4) from biosynthetic method CH4 Molar Mass CalculationĬH4 has a molecular mass of 16.04 g/mol, which may be computed as follows. These reactions are called enzymatic reactions. Initially, car5bon dioxide reacted with hydrogen gas formed methane in the presence of methyl coenzyme M reductase. The outcome product is methane also in the gas phase. It is the gas phase reaction between, carbon dioxide in the gas phase, and hydrogen in the gas phase. 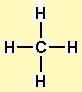
It is created primarily through methanogenesis. Methane(CH4) is a colorless gas with an odorless form.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
